Recycling is good for many things. However, when bogus concepts in fertilizer use are recycled, it can be expensive for farmers. One example is the idea that there are “ideal” ratios of calcium (Ca) to magnesium (Mg) to potassium (K) in soils. This “ideal ratio” concept, once thought to have disappeared from production agriculture, is being promoted by some for use in 2016. It’s being recycled. This is a concept that should not be used. To understand the basis for this statement, some explanation of the fundamentals is needed.
There are negative electrical charges in all soils. These charges are associated primarily with the clay sized particles. The number of these charges can be measured by laboratory analysis and is reported by as the Cation Exchange Capacity (CEC). This measurement is reported as milliequivalents per 100 grams of soil. These negative electrical charges are also referred to as exchange sites. The CEC varies with soil texture as shown in Table 1.
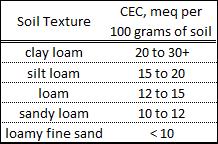
Positively charged nutrients (ammonium, calcium, magnesium, potassium, and micronutrients except for boron) are loosely held by the negative charges. This discussion will focus on the relationship of CEC to Ca, Mg, and K. It will examine the relationship, if any, between the ratios of these nutrients and crop production.
The relationships among nutrients held by soils and crop productivity was first researched by Dr. Bear and co-workers in 1945 using beach sand in New Jersey. They measured crop yields and nutrients held on the exchange sites. From these measurements, they concluded that an “ideal soil” should have 65% of the exchange sites occupied by Ca. Likewise, they stated that 10% should be occupied by Mg and 5% by K. These percentages are, then, converted to a ratio of 13:2:1 for Ca, Mg, and K respectively. Therefore, the “ratio” concept was born. Because of interest in liming at the time, there was more interest in the Ca:Mg ratio reported as 6.5:1.
The concept of nutrient ratios was further researched by Dr. Graham at the University of Missouri. He published research which relaxed the rigid ratios published by Dr. Bear and others. Dr. Graham wrote that the percentages of nutrients on exchange sites could be in the range of 65% to 85% for Ca, 6% to 12% for Mg, and 25 to 5% for K. The research of Dr. Graham was not given serious consideration and the 6.5 to 1 ratio of Ca to Mg persisted. This ratio was promoted and used to sell lime and/or magnesium fertilizers. If the ratio was too high, it could be reduced by the use of magnesium fertilizers. If the ratio was too low, lime was needed.
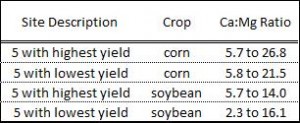
Over the years, the effect of the Ca:Mg ratio on crop yield has been studied by faculty at Land Grant Universities in the Corn Belt. Some of the most active research was conducted at Ohio State University. The research was conducted at 20 0n-farm locations in each of two years. The results are summarized in Table 2. They show that the ratio of Ca:Mg was not at all related to the yield of corn or soybeans. Research conducted in Nebraska also showed that the ratios for Ca:Mg or Ca:K or Mg:K had no effect on crop production.
The Ca:Mg ratios in Minnesota vary widely across the state. In western Minnesota where exchangeable Ca is high, there is a high Ca:Mg ratio. Yet, the addition of Mg to a fertilizer program has no effect on yield. In eastern Minnesota, there are situations where the use of Mg in a fertilizer program has increased yield. The need for Mg in a fertilizer program is based on a soil test for that nutrient — not he ratio with Ca.
The conclusions of research conducted by several universities for several years are consistent. Fertilizer recommendations should not be based on the ratio of one nutrient to another. Use of the “ideal ratio” concept is a waste of money. Dr. Charles Black of Iowa State University provided a good summary. He wrote: “the Values suggested by Bear, Pierce and Malcolm (1945), Graham (1959), and Amacher (1981) all represent a good educational guess regarding satisfactory exchangeable cation ratios in most soils for most crops. The fact is, though, that research has not documented that there is an optimum exchangeable cation ratio for any crop”.
- Value of Soil Organic Matter - March 8, 2017
- Movement of Nutrients from Soil to Plants - September 20, 2016
- Understanding the Basics of Water in Soils - August 14, 2016
